
Minoryx Therapeutics completes enrollment in FRAMES phase 2 trial with leriglitazone in Friedreich’s Ataxia
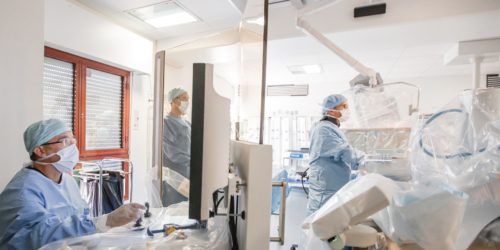
Robocath successfully carries out its first robotic coronary angioplasties in humans

Imcyse to present first-in-human phase 1b study results in type 1 diabetes at EASD 2019

Imcyse to present positive results in EAE model of multiple sclerosis at ECTRIMS 2019

Imcyse reports successful first-in-human Phase 1b study in type 1 diabetes with IMCY-0098

GamaMabs Pharma announces presentation of murlentamab phase 2 study results in colorectal cancer at ESMO World Congress on Gastrointestinal Cancer

Minoryx Therapeutics announces first patient dosed in the FRAMES phase 2 trial in Friedreich’s Ataxia

GamaMabs Pharma to present updated results at 2019 ASCO Annual Meeting from the First-In-Human clinical study of murlentamab in advanced gynecological cancers

